Exceptional Clinical Efficacy & Safety
Since the “Stem Cell Therapy for Knee Osteoarthritis” project landed in Boao Lecheng this year, over 300 patients have successfully completed their injection treatments.
The results demonstrate that just 4 weeks after the stem cell injection, patients experienced comprehensive improvements across all functional indicators:
- Symptom Relief: Significant reduction in joint pain and a decrease in joint stiffness.
- Mobility Restored: Patients gradually regained their ability to stand, walk, climb stairs, go shopping, and perform daily housework.
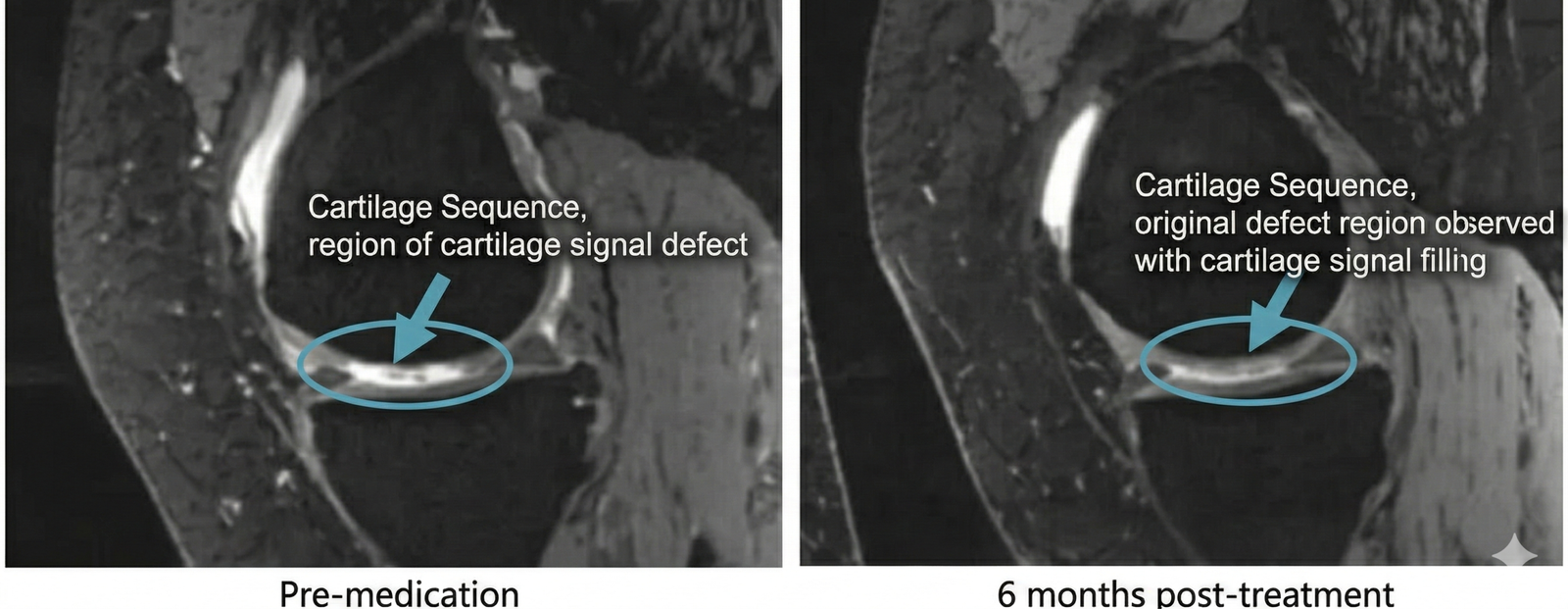
- Imaging Proof: MRI examinations revealed alleviated knee edema and a verified increase in articular cartilage volume.
- Flawless Safety: Zero serious drug-related adverse events have occurred.
These clinical outcomes prove that stem cell therapy is an effective treatment capable of not only improving or curing knee osteoarthritis but demonstrating excellent safety. It is a powerful therapy that can halt and even reverse the pathological progression of knee osteoarthritis.

Case Study 1
Margaret: Returning to the Garden
Patient Profile
Margaret, 65 years old. A retired middle school music teacher from Chicago, USA. A formerly active woman who loved hiking and gardening, her life was overshadowed by a 12-year battle with bilateral knee osteoarthritis (KL Grade III).
The Dilemma
She relied heavily on strong painkillers and required a walker just to leave the house. She could no longer attend her beloved community choir rehearsals, and even walking down to her garden bushes became an impossible luxury. Local doctors strongly recommended total knee replacement (TKR) surgery, but her fear of the operation kept her hesitant.
The Turning Point & Treatment
After researching China’s advancements in stem cell therapy, she flew halfway across the globe to our clinic. In August 2023, Margaret received Autologous Adipose-Derived Stem Cell (ADSC) intra-articular injections. Her own adipose tissue was cultured in our lab, delivering 100 million (1×10⁸) cells per infusion. The procedure was minimally invasive and entirely avoided the painful recovery of traditional surgery.
Recovery Story
“For years, I dreaded my grandchildren’s birthday parties because it meant sitting in a wheelchair all day. Now, not only can I run and jump, but I am back to my gardening. I’ve said goodbye to those awful painkillers and saved my natural joints. Thank you for this cross-border miracle.”

Core Clinical Data Tracking
| Metric | Baseline | 3 Months | 1 Year |
|---|---|---|---|
| VAS Pain Score (0-10) | 8 | 2 | 1 |
| WOMAC Function Score | 78 | 39 (↓ 50%) | 32 (↓ 59%) |
| Range of Motion | Flexion 85° | Flexion 125° (↑ 47%) | Flexion 135° (Normal) |
| MRI Cartilage Thickness | 1.2 mm | 1.8 mm (↑ 50%) | 2.0 mm (↑ 67%) |

Case Study 2
Liam: The Lightning Return to the Court
Patient Profile
Liam, 25 years old. A professional basketball point guard from London, UK. Currently in his prime, a severe under-the-basket collision brought his career to a sudden halt.
The Dilemma
Diagnosed with a Grade III meniscus tear and Grade II cartilage damage in his left knee. After 6 months of local physical therapy with minimal improvement, specialists suggested a partial meniscectomy. However, for a pro athlete, losing meniscus tissue often means a permanent loss of explosive power and early retirement. “If I don’t operate, I can’t play. If I operate, I may never return to my peak.”
Treatment Strategy: UC-MSC Protocol
Determined to save his career, Liam chose our non-surgical biological repair protocol in March 2024. He received Allogeneic Umbilical Cord Mesenchymal Stem Cells (UC-MSC). The dosage was 10 million cells per session, split into 2 sequential injections spaced 4 weeks apart. UC-MSCs possess extraordinary immunomodulatory and regenerative properties, capable of repairing damaged meniscus fibrous tissue and cartilage matrix.
Recovery Story
“People thought taking this trip to China was a massive risk. But by month 3, I was doing pain-free basic training. By month 6, I was back on the official match roster! The biggest shock was the MRI—my doctor pointed out that the torn meniscus was regenerating and the cartilage was smooth again. I didn’t have to cut out a single piece of my tissue. This trip didn’t just heal my knee; it saved my career.”
Core Clinical Data Tracking
| Metric | Baseline | 3 Months | 6 Months |
|---|---|---|---|
| VAS Pain Score | 9 | 3 | 1 |
| Lysholm Knee Score | 45 | 78 | 92 (Normal) |
| Meniscus GAG Content | 1.2 (Normal: 5.02) | 2.5 | 3.36 (67% Recovered) |
| Meniscus Volume | 22% Atrophy | 12% Growth | 18% Growth |
| Joint Effusion | Massive | Mild | Fully Absorbed |
Case Study 3
Thomas: Freedom from the Scaffolding
Patient Profile
Thomas, 45 years old. A senior civil engineer from Berlin, Germany. For Thomas, his knees are the pillars of his career, requiring constant navigation through construction sites and scaffolding.
The Dilemma
Three years ago, Thomas underwent a “partial meniscectomy” locally for a severe tear. The relief was short-lived; within a year, persistent swelling returned. His knees felt like rusted machinery, generating agonizing pain during squats and stairs. Facing a long-term medical leave crisis and the grim diagnosis of “nothing left but another surgery,” he sought regenerative alternatives globally.
Treatment Strategy: The Regenerative “Golden Duo”
In May 2024, our expert panel administered a precision injection therapy combining Autologous Adipose-Derived Stem Cells (ADSC) with Platelet-Rich Plasma (PRP). The ADSCs provided the “repair seeds” while PRP offered the “growth soil,” dramatically boosting regeneration efficiency. Utilizing high-resolution ultrasound guidance, the cells were delivered directly to the degenerated cartilage edges over 4 intensive sessions.
Recovery Story
“In Germany, they told me post-surgical wear was inevitable. But in China, a miracle happened. After the first month, the swelling I endured for two years completely vanished. By month 3, I started weight-bearing activities without a single ‘protest’ from my knee. It’s been a year—I’m back on the construction site and even practicing Tai Chi every morning. Regaining my health without enduring another painful surgery feels absolutely incredible!”

Core Clinical Data Tracking
| Metric | Baseline | 1 Month | 6 Months | 1 Year |
|---|---|---|---|---|
| VAS Pain Score | 7 | 3 | 1 | 0 |
| Range of Motion | Flexion 90° | Flexion 110° | Flexion 130° | Flexion 135° |
| Swelling Level | Severe (+3cm girth) | Mild (+0.8cm) | No Swelling | No Swelling |
| MRI Evaluation | Meniscus edge defect, Synovial hyperplasia | Defect shrinking, Inflammation reduced | New tissue covering defect | Stable structure, No recurrence |

Case Study 4
Elena: A New Life Without Pain
Patient Profile
Elena, 59 years old. An elegant art gallery curator from Paris, France. Her passion for art and life was nearly entirely stripped away by 9 years of degenerative bilateral knee osteoarthritis (KL Grade III).
The Dilemma
Every step was exhausting; her knees felt like dry wood violently rubbing together. Severe joint swelling forced her to literally “hold onto the walls” to navigate stairs. European specialists offered only one solution: Total Knee Replacement (TKR). Terrified of the foreign body sensation and potential surgical complications, Elena desperately wanted to preserve her natural joints.
Treatment Strategy & The Journey East
In the fall of 2023, after learning about China’s mature clinical experience with Adipose-Derived Stem Cells, she crossed the Eurasian continent to find her last hope at our clinic.
Recovery Story
“Before coming to China, I thought I would spend the rest of my life clinging to walls and swallowing painkillers—it was my darkest time. Three months after treatment, the burning pain that haunted me for thousands of days and nights vanished. I no longer rely on any medication. I can jog with my grandson in the park, swim, and even attempt short hikes. Looking at the smooth, regenerated cartilage on my MRI, I couldn’t believe it. I saved my joints without a cold metal replacement. This happiness of regained freedom is something money simply cannot buy.”
Core Clinical Data Tracking
| Metric | Baseline | 3 Months | 6 Months |
|---|---|---|---|
| VAS Pain Score | 8.2 | 2.1 | 1.5 |
| Range of Motion | Flexion 70° | Flexion 135° (↑ 93%) | Flexion 140° |
| MRI Cartilage Thickness | 0.9 mm | 1.7 mm (↑ 89%) | 2.0 mm (↑ 122%) |
| Walking Ability | Needs wall support, <500m | Independent walking >2000m | Normal walking & stairs |
Ready to Start Your Recovery Journey?
Avoid invasive replacement surgeries and discover what regenerative medicine can do for your joints.
Professional Knee Stem Cell Protocol
Connect with our international medical team to create your personalized treatment plan at our Boao Lecheng facility.
View Detailed Pricing & Book